 However, enthalpies usually provide a very good starting point. To determine whether a reaction will actually occur under a given set of conditions, the important value to check is Gibbs Free Energy. The balanced equation for when pentane gas (C5H12) combusts with oxygen gas (O2) to form water (H2O) and carbon dioxide (CO2) is CH + 8O -> 5CO + 6HO. It always produces carbon dioxide and water vapor. Note that my calculations all centre around enthalpy. A combustion reaction occurs when a compound that contains carbon and hydrogen is burned in Oxygen gas (O2). 
The key takeaway is, however, that the reaction as proposed is endothermic and you need some sort of driving force (something more exothermic) to make it happen. carbon dioxide (CO2) and carbon monoxide (CO). More complicated systems are evidently possible, the reactions and equations will just get more and more complicated. gas equation of state in terms of the universal gas constant as follows. As water would be produced by adding hydrogen gas to the reaction, the reactant side only gains another zero. 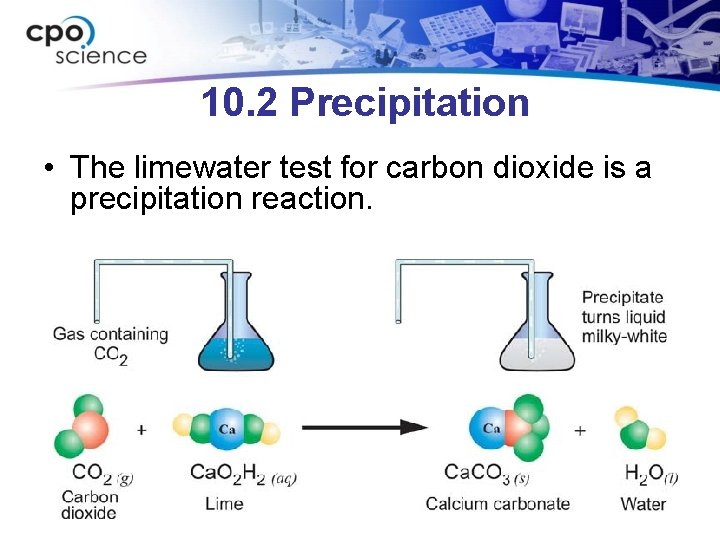
From a table of values on Wikipedia, we find in the row for carbon dioxide that this reaction has an enthalpy change of $\mathrm$ according to the same Wikipedia article linked above. 1, carbon monoxide + oxygen carbon dioxide 2, CO + O 2 CO 3, Reactants: 1 × C, (1 × O) + (2 × O) 3 × O. We can look up the enthalpy change associated with it. 
This reaction is simply a bond dissociation (specifically, a carbon-oxygen covalent double bond is broken). Unfortunately, the question as stated is thermodynamically impossible. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
